|
Why do halogens get more reactive going upwards in group 7? The outer electron is more easily transferred to say an oxygen atom, which needs electrons to complete its full outer shell. The more electron shells (rings) between the nucleus and outer electron also creates shielding and again this weakens the nuclear attraction. The distance "c" is greater than "a" and the force of attraction between the nucleus and the outer shell (rings) diminishes with distance. Why do alkali metals get more reactive going down group 1?Īlkali metals from lithium to potassium get more reactive because the force of attraction between the nucleus (core) and the outer electron gets weaker as you go down group 1 elements. The following is a reactivity chart found on the Internet.As you go down group 1 (the alkali metals) in the periodic table, the elements get more reactive.Īs you go up group 7 (the halogens), again, the elements become more reactive.No, I wouldn't expect this element to be found in an unreacted elemental form because based on the information it fits the violently reactive section. Would oyu expect this metal to be found in its unreacted elemental form? Explain In my opinion by looking at the periodic table visually and based off of the previous questions I think Francium is the most reactive element out of the ones that were given. Which is the most reactive metal in the periodic table? Explain.Arrange each set of the following metals in order from least to most reactive using conclusions you just made.What can you conclude about the reactivity of metals as you move across a period? When you travel from left to right across a period in this case the reactivity of the elements tend to decrease. 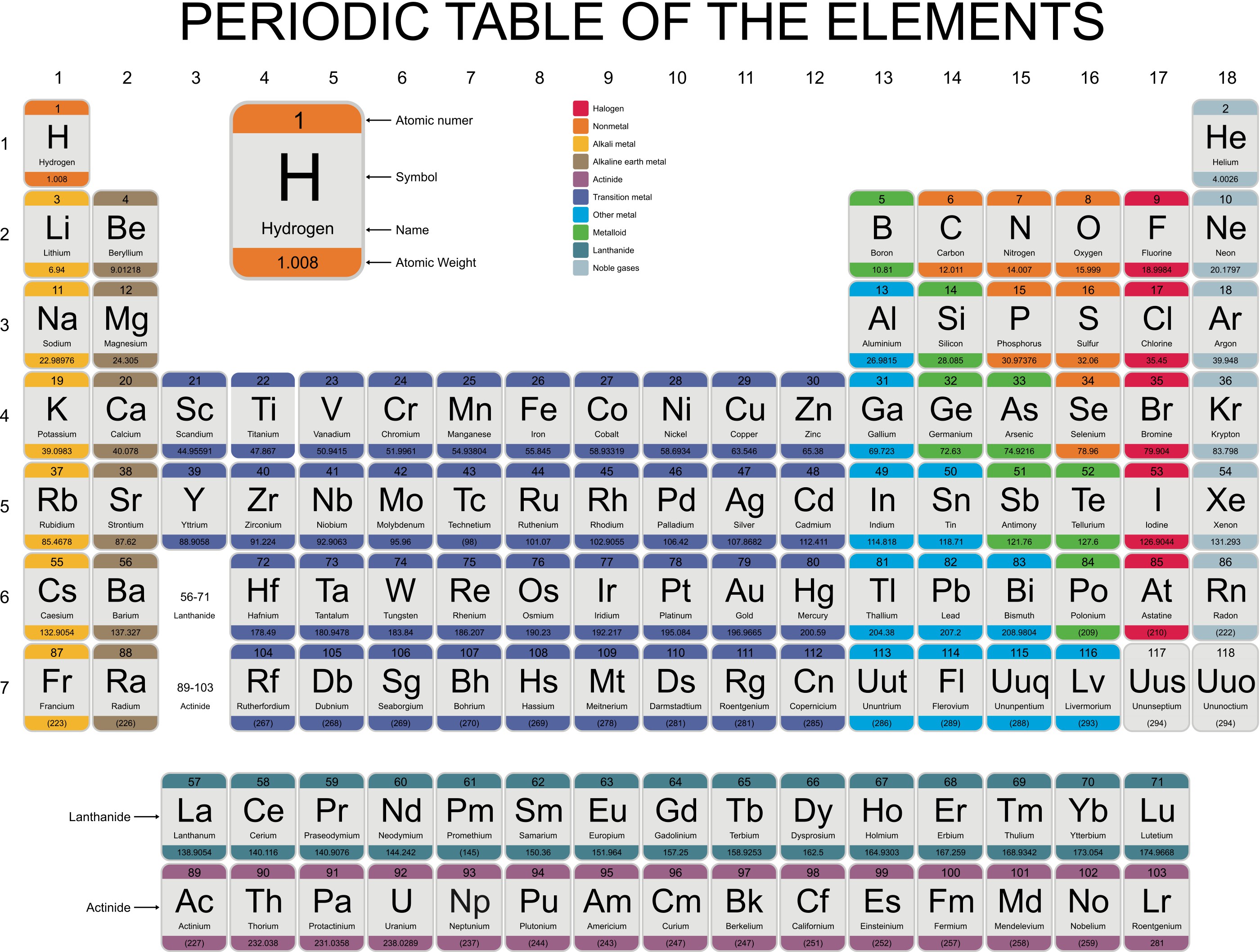
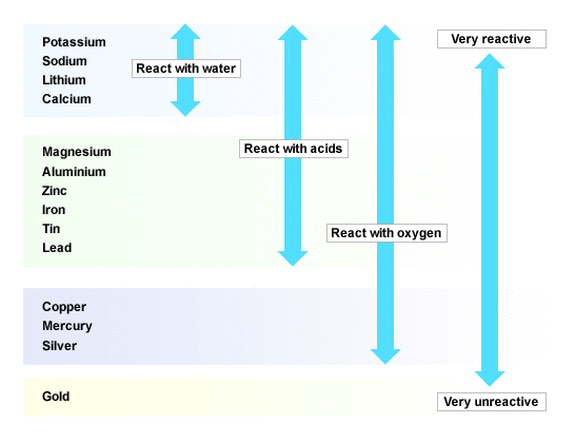
However, Alkali metals are known to be more violently reactive than the Alkali earth metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
